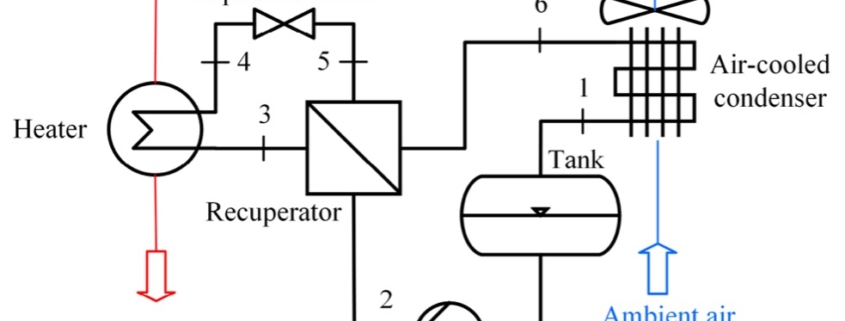
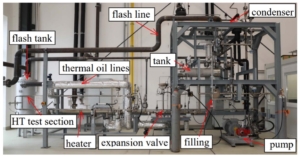
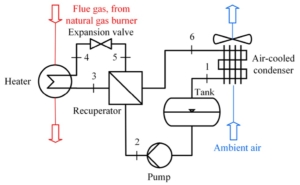
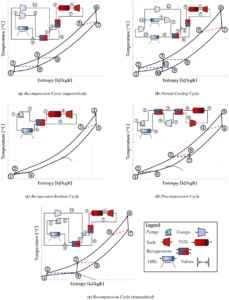
Paper on aerodynamic and mechanical design of large gas turbines presented by City, University of London and Baker-Hughes at ASME Turbo Expo, Boston (USA)
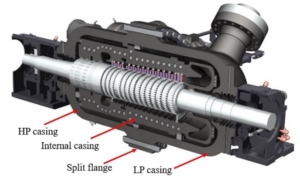
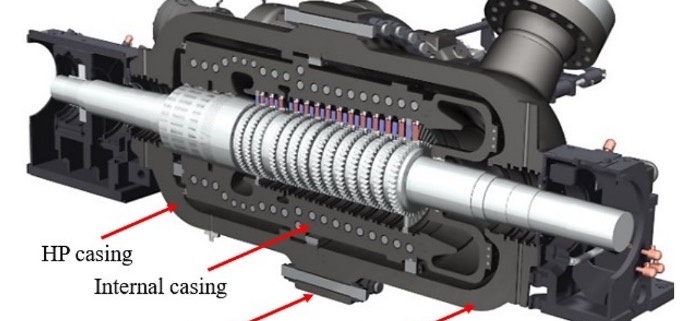
Turbine assembly cross section (©Baker Hughes Company, All Rights Reserved)
The results of the collaboration between City, University of London (United Kingdom) and Baker-Hughes (Italy) have been have presented at ASME Turbo Expo in Boston (USA). This joint research has taken place within Work Package 3 – Turbomachinery Design of SCARABEUS, aimed at developing turbine designs able to attain high efficiency when working with Carbon Dioxide mixtures at very high pressures and temperatures.
Turbine design is strongly influenced by the composition of the working fluid because of the impact of this feature on the operating conditions of the cycle that attain peak thermal efficiency, and also the impact of composition on fluid characteristics.
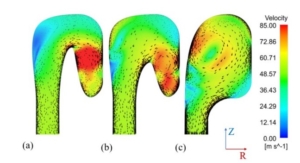
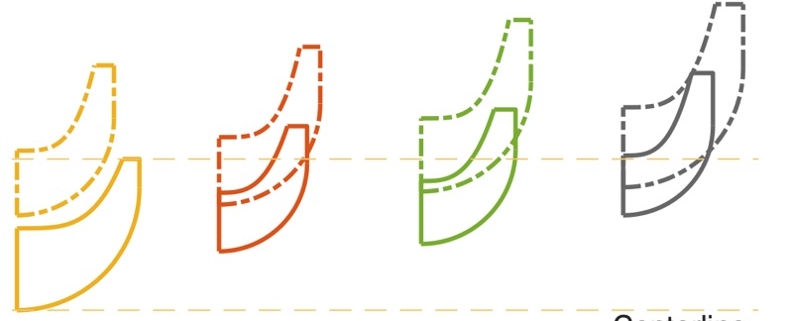
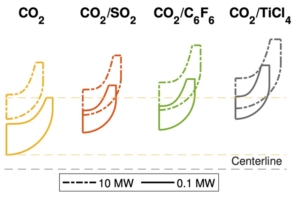
Flow field of three different designs of the exhaust section
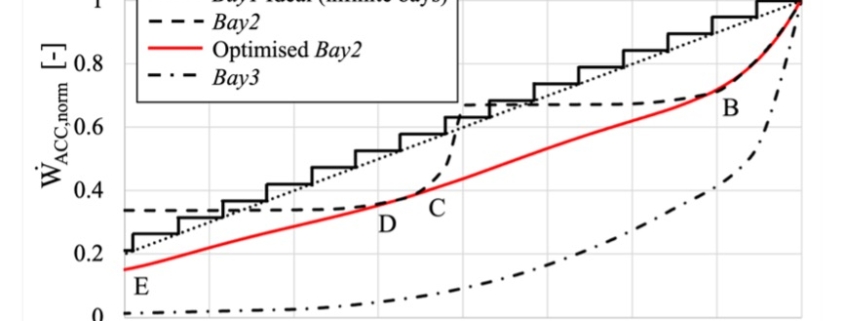
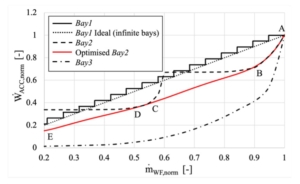
The paper presents the results of the unsteady simulations of the last turbine stage and exhaust section to assess unsteady loads on the rotor, as well as aerodynamic losses in the diffuser and exhaust section. Rotordynamics are also studied.
The paper can be downloaded free of charge from the conference website (link). Check the abstract below:
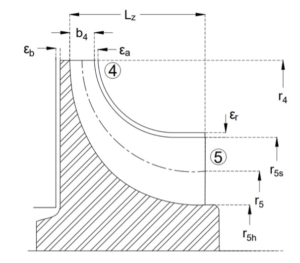
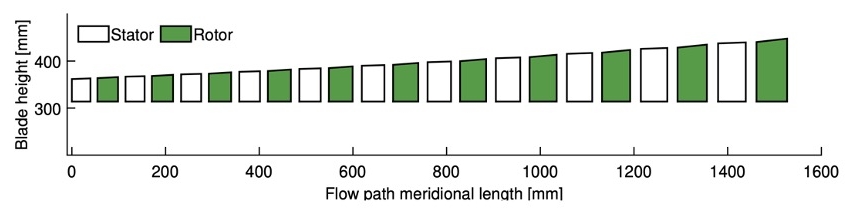
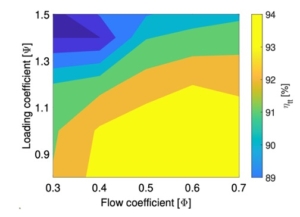
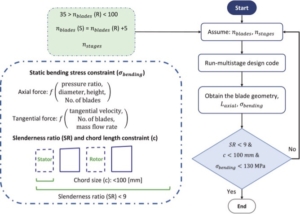
In this paper, the design of a large-scale axial turbine operating with supercritical carbon dioxide (sCO2) blended with sulfur dioxide (SO2) is presented considering aerodynamic and mechanical design aspects as well as the integration of the whole turbine assembly. The turbine is 130 MW, designed for a 100 MWe concentrated-solar power plant with turbine inlet conditions of 239.1 bar and 700 °C, total-to-static pressure ratio of 2.94 and mass-flow rate of 822 kg/s. The aerodynamic flow path, obtained in a previous study, is first summarised before the aerodynamic performance is evaluated using both steady-state and unsteady 3D numerical models to simulate the aerodynamic performance of the turbine. Whole-annulus unsteady simulations are performed for the last turbine stage and the exhaust section to assess the unsteady loads on the rotor due to downstream pressure field distortion and to assess aerodynamic losses of the diffuser and exhaust section. The potential low engine order excitation on the last rotor stage natural frequency modes due to downstream pressure distortion is assessed. The design of the turbine assembly is constrained by current manufacturing capabilities and the proposed working fluid properties. High-level flow-path design parameters, such as pitch diameter and number of stages, are established considering a trade-off between weight and footprint, turbine efficiency and rotordynamics. Rotordynamic stability is assessed considering the high fluid density related to cross coupling effects. Finally, shaft end sizing, cooling system design and the integration of dry gas seals are discussed.